| Monotard | |
|---|---|
| Lente by Novo Nordisk | |
| intermediate-acting | |
| r-DNA/GE/GM | |
| U40, U100 | Zinc |
Action in dogs:
| |
| Line: Novo | |
| Also known as: Novolin L | |
| Similar to: Huminsulin L, Huminsulin Long, Humutard, Humulin Lente, Humulin L Humulin Lenta, Humulina L, Humuline Long Umuline Lente, Umuline Zinc Compose Names of Lilly r-DNA/GE/GM insulins worldwide | |
| Use and Handling: | |
| Shelf Life: 30 months | Type: cloudy |
| When Opened: 6 weeks room temp. | |
| In Pen: N/A | |
| Notes: Protect from light and heat Do Not Freeze, Resuspend Do not use if product does not re-suspend Do not use intravenously [1] store at 2-8C | |
Novo Nordisk Monotard HM--GE Human Lente Insulin.
|
Note that some countries have the brand name in U40 strength. | ||
Monotard was Novo Nordisk's Euro brand name for their U100 lente-type insulin [2][3][4] which is intermediate-acting. As of early 2006, this insulin was discontinued. [5][6]
It was known as Novolin L [7] in North America; Eli Lilly made a comparable product, Humulin L, discontinued in the EU/UK in July 2004, [8] also discontinued in North America in July 2005. [9][10] Lilly's version of the insulin was also known as Umuline Zinc Compose, Humulina L and Humuline Long.
Like all Lente insulins, [11] regardless of species or strength, Monotard was a suspension of crystalline (ultralente) and amorphous (semilente) insulin in a ratio of 7 crystalline parts to 3 amorphous parts. [12]
Why Lente doesn't equal 70/30[]
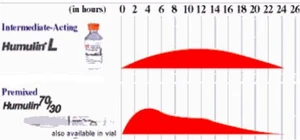
Direct comparison of insulin activity profiles for Lente and 70/30 insulins. The early strong action from the non-suspended R/neutral/normal insulin is seen at the "bump" from hours 0-6.
Let's look at the differences in the two insulins. Lente is comprised of 70 % long-acting Ultralente insulin and 30% short-acting Semilente insulin. So proportions of a long-acting and short-acting insulin are being combined to produce a intermediate-acting insulin. Both Ultralente and Semilente insulin are suspended by adding zinc and the size of their respective insulin crystals. The largest insulin crystals are those of Ultralente, while Semilente contains the smallest, or microcrystals. Simply put, you're combining a long insulin and a short insulin to make an intermediate-acting one.[13]
70/30 insulin starts with 70% NPH/isophane insulin, an intermediate-acting insulin which is suspended by protamine. To that, 30% R/neutral/normal insulin, which is short-acting and soluble, meaning there is no suspension; nothing is added to this insulin to delay its action. In this case, you are taking an intermediate-acting insulin with a suspension and adding to it a short-acting one with no suspension to create an intermediate-acting insulin.
So we have an insulin (Lente), made up of a long-acting and short-acting insulin, both with zinc suspensions, and a mixed insulin (70/30), made from an intermediate-acting protamine suspended insulin and a short-acting one without any suspension. Because of these differences, the insulin action profile for these two insulins is going to be quite different, even though both insulins are classed as intermediate-acting.
What Lente Is Not[]
No Lente-type insulin regardless of species can contain any NPH/isophane insulin [14] or any R/Neutral insulin. [15][16]
Both are chemically impossible: the phenol preservative present in NPH/isophane alters the action of Lente-type insulins, creating a mixture with an approximate action of R/Neutral. [17][18]
The zinc suspension of Lente-type insulin binds R/Neutral, causing the short-acting insulin to slow, losing its short-acting effect. [19][20]
Before the invention of VetPen, Lente-type insulins could not be dispensed in pen or cartridge form because the glass ball formerly used to mix the insulin in these devices shattered the Lente crystals.[21]
Combining Lente Family Insulins[]
|
None of the Lente family of insulins (semilente, Lente, Ultralente) can be combined with [17] NPH/isophane insulins. The phenol preservatives present in NPH-type insulins alters the Lente-types to the point where they become a close approximation of R/neutral, with regard to action. [22][23] Keeping the phenol preservatives in mind, all protamine-suspended insulin mixes would be "off limits" regarding same syringe mixing with any Lente-type insulins. [23] | ||
Insulin manufacturers [24] indicate that R/neutral and semilente, Lente, ultralente insulins are able to be combined in the same syringe, but only just before injection. In pre-filled syringes, the zinc suspension of the Lente-type insulins binds the R/neutral, causing it to lose its short-acting effect. Various studies have documented this, and some doctors advise against using R/neutral in the same syringe with the Lente family of insulins. [16][25][26][23]
|
Lente Insulins | |
|---|---|

|
Intermediate acting Non-soluble |
| Betasint Bovine Lente Hypurin Bovine Lente | |
| Iletin Lente (No longer produced.) Beef Lentard (No longer produced.) | |
| Iletin I Lente (No longer produced.) [27] Lentard MC (No longer produced.) | |
| Betasint Porcine Lente, Lente CHO | |
|
Iletin II Lente (No longer produced.) [28] | |
| Monotard, Novolin L (No longer produced.) Huminsulin L, Huminsulin Long Humutard, Humulin L Humulin Lenta, Humulin Lente Humulina L, Humuline Long Umuline Lente, Umuline Zinc Compose (No longer produced.) | |
The following are lente type zinc suspension insulins but since they are produced for use in animals, they are not found in BNF, but in its sister veterinary formulary.
|
Lente Insulins | |
|---|---|

|
Intermediate acting Non-soluble |
| Insuvet Lente (No longer produced.) | |
| Caninsulin, Vetsulin | |
These fall into the category of intermediate-length insulins.
The Novo insulins, Ultratard and Monotard, were available slightly longer in Europe. [6]
From the announcement: Monotard and Ultratard will not be available in the UK after February 2006. The discontinuation of these insulins was initially announced in September 2004. Initially the discontinuation was scheduled for February 2006, however this date has now been brought forward to October 2005.
References[]
- ↑ Maddison, Jill E.,Page, Stephen W.,Church, David B. (2008). Small Animal Clinical Pharmacology. Saunders Ltd..
- ↑ Prescribing Novo Insulins.
- ↑ Information on Monotard. Novo Nordisk.
- ↑ Definition of Lente Insulin. Merck Manual.
- ↑ Photo of Monotard Insulin.
- ↑ 6.0 6.1 Diabetes UK Monotard/Ultratard Discontinued. Diabetes UK (2005). Cite error: Invalid
<ref>tag; name "Discontinue" defined multiple times with different content - ↑ Novolin L Discontinuation Notice. US Food and Drug Administration (2003).
- ↑ Discontinuation of Humulin L. Diabetes UK (July 2004).
- ↑ Lilly EU/UK Product Sheet & Time Activity Profiles. Eli Lilly.
- ↑ Humulin L Discontinuation Notice. US Food and Drug Administration.
- ↑ Scientific Discussion-Monotard. EMEA.
- ↑ Dumitriu, Severian (2001). Polymeric Biomaterials, Revised and Expanded 1104. CRC Press.
- ↑ Greco, Deborah (2010). Treating Diabetes Mellitus in Dogs and Cats. Western Veterinary Conference.
- ↑ Combining Lente-type Insulins with Phenol-Preserved Insulins. National Federation for the Blind.
- ↑ Lente Zinc Suspension Causes Loss Of R/Neutral Short-Acting Effect. Endotext.org.
- ↑ 16.0 16.1 Huffman DM, Garber AJ. (1991). Availability of Soluble (R/Neutral) Insulin in Mixed Preparations With Crystalline (Lente) & Ultralente GE Insulin. Clinical Therapeutics. Cite error: Invalid
<ref>tag; name "Huffman" defined multiple times with different content - ↑ 17.0 17.1 Lente-Type Insulins & NPH/Isophane Insulins-A Bad Combination. National Federation for the Blind. Cite error: Invalid
<ref>tag; name "Bad" defined multiple times with different content - ↑ Havlik I, Galasko G, Alberts E, Furman KI, Seftel HC. (1988). Solubility Changes on Mixing Short- and Long-acting Insulin Preparations. South African Medical Journal.
- ↑ Deckert, T. (1980). Intermediate-Acting Insulin Preparations: NPH (Isophane) & Lente. Diabetes Care.
Note--in 1980, there were no r-DNA/GE/GM insulins - ↑ Resource Guide. American Diabetes Association (2005).
- ↑ Hanas, Ragnar (1999). Insulin-Dependent Diabetes-Page 10. ChildrenWithDiabetes.
- ↑ Lente-Type Insulins & NPH/Isophane Insulins-A Bad Combination. National Federation for the Blind.
- ↑ 23.0 23.1 23.2 Insulin Therapy-Mixing Precautions. RxEd.org. Cite error: Invalid
<ref>tag; name "Rx" defined multiple times with different content - ↑ Insulin Producers vs Doctors Re:Combining R/Neutral & Lente-type Insulins. Endotext.org.
- ↑ Bilo HJ, Heine RJ, Sikkenk AC, van der Meer J, van der Veen EA. (1987). Absorption Kinetics & Action Profiles-Single Subcutaneous Administration of Human Soluble (R/Neutral) & Lente Insulin. Diabetes Care.
- ↑ Heine RJ, Sikkenk AC, Eizenga WH, van der Veen EA. (1983). Delayed Onset of Action of Soluble (R/Neutral) Insulin After Premixing With Lente Insulin Diabetes. Research & Clinical Practice.
- ↑ Iletin I Lente-Beef/Pork Insulin Shown at Right.
- ↑ Carton Image-Iletin II Lente.
More Information[]
- Converting a Diabetic Pet from a Human Lente Insulin Preparation to Caninsulin Intervet
- No More Humulin Lente-What Do We Use? Richter, Keith, ACVIM
- NACDS-Insulin Chart-Page 2
- Insulin therapy for dogs and cats Dowling, Patricia, September 1995, Canadian Veterinary Journal
A discussion of Lente insulins.
- Lente Insulins-Injectable Suspensions West Virginia University College of Pharmacy-2009
- Insulin human zinc (Lente)
 |
 |
 |